 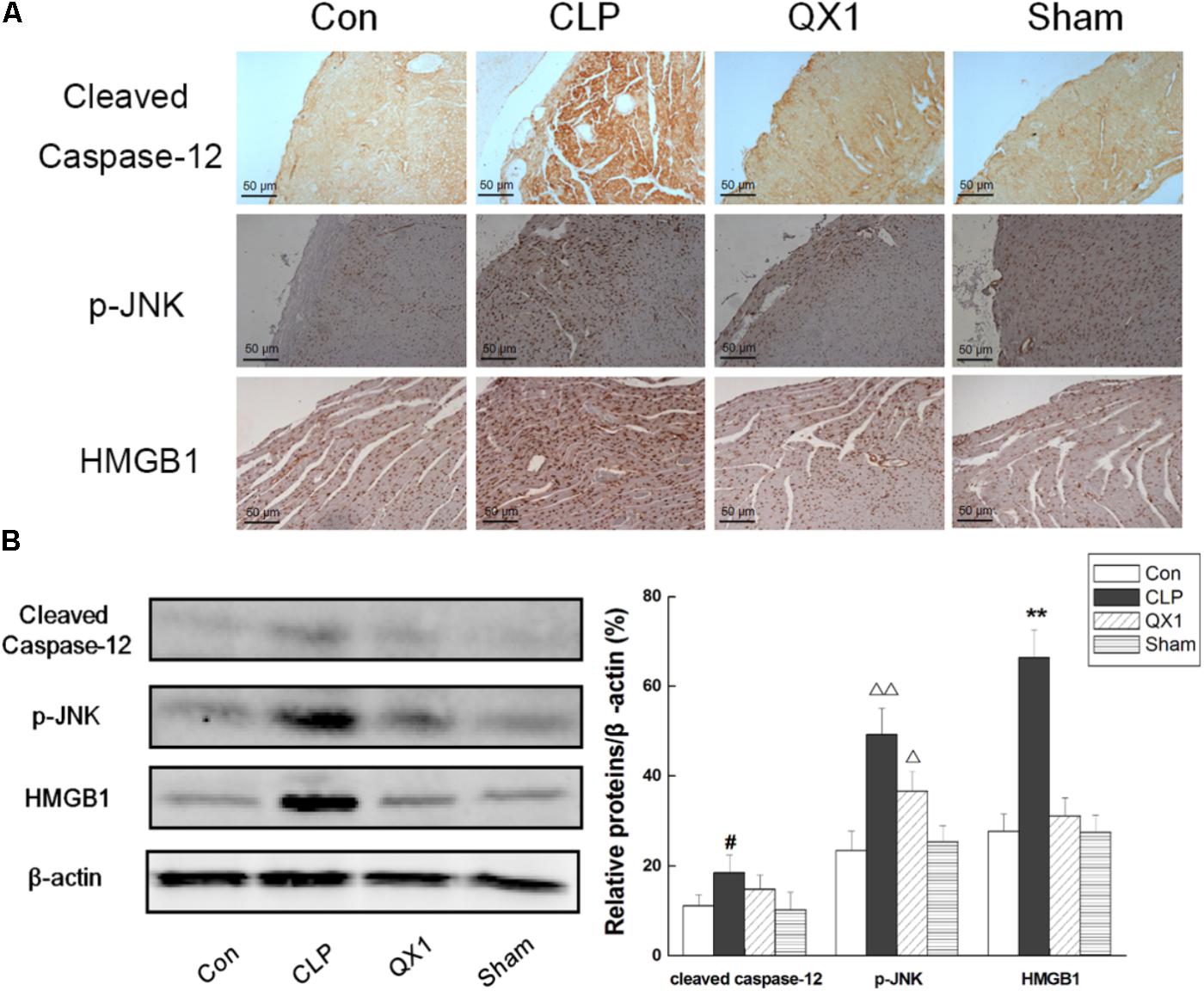
Tricine–SDS-PAGE is also used preferentially for doubled SDS-PAGE (dSDS-PAGE), a proteomic tool used to isolate extremely hydrophobic proteins for mass spectrometric identification, and it offers advantages for resolution of the second dimension after blue-native PAGE (BN-PAGE) and clear-native PAGE (CN-PAGE). These lower concentrations facilitate electroblotting, which is particularly crucial for hydrophobic proteins. The concentrations of acrylamide used in the gels are lower than in other electrophoretic systems. It is the preferred electrophoretic system for the resolution of proteins smaller than 30 kDa. Tricine–SDS-PAGE is commonly used to separate proteins in the mass range 1–100 kDa. eCLIP enables integrative analysis of diverse RBPs to reveal factor-specific profiles, common artifacts for CLIP and RNA-centric perspectives on RBP activity. We generated 102 eCLIP experiments for 73 diverse RBPs in HepG2 and K562 cells (available at ), demonstrating that eCLIP enables large-scale and robust profiling, with amplification and sample requirements similar to those of ChIP-seq. 
By simplifying the generation of paired IgG and size-matched input controls, eCLIP improves specificity in the discovery of authentic binding sites. We have developed an enhanced CLIP (eCLIP) protocol that decreases requisite amplification by ∼1,000-fold, decreasing discarded PCR duplicate reads by ∼60% while maintaining single-nucleotide binding resolution. 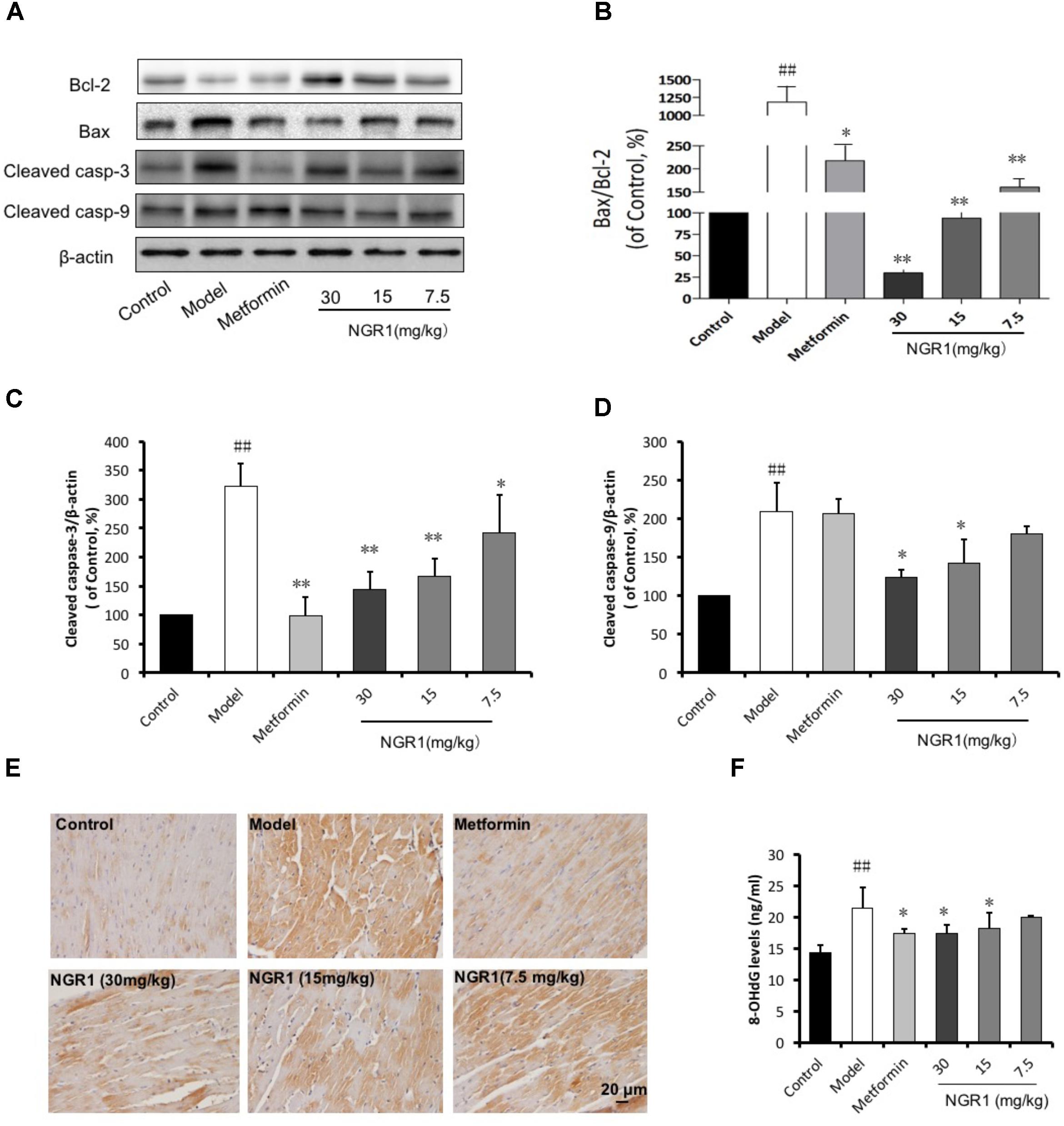
However, current CLIP protocols are technically demanding and yield low-complexity libraries with high experimental failure rates. lessĪs RNA-binding proteins (RBPs) play essential roles in cellular physiology by interacting with target RNA molecules, binding site identification by UV crosslinking and immunoprecipitation (CLIP) of ribonucleoprotein complexes is critical to understanding RBP function. Here we describe basic protocols to assay for endogenous LC3-II by immunoblotting, immunoprecipitation, and immunofluorescence. Thus, lysosomal turnover of the autophagosomal marker LC3-II reflects starvation-induced autophagic activity, and detecting LC3 by immunoblotting or immunofluorescence has become a reliable method for monitoring autophagy and autophagy-related processes, including autophagic cell death. At the same time, LC3-II in autolysosomal lumen is degraded. Autophagosomes fuse with lysosomes to form autolysosomes, and intra-autophagosomal components are degraded by lysosomal hydrolases. Concomitantly, a cytosolic form of LC3 (LC3-I) is conjugated to phosphatidylethanolamine to form LC3-phosphatidylethanolamine conjugate (LC3-II), which is recruited to autophagosomal membranes. During autophagy, autophagosomes engulf cytoplasmic components, including cytosolic proteins and organelles. Microtubule-associated protein 1A/1B-light chain 3 (LC3) is a soluble protein with a molecular mass of ∼17 kDa that is distributed ubiquitously in mammalian tissues and cultured cells. Both approaches can be completed in a day. The latter protocol has been optimized to allow an increased throughput, as potential applications require large numbers of samples. One approach involves sample workup and detection using quantitative western blotting, whereas the second is performed directly in solution and relies on the induced proximity of two target-directed antibodies upon binding to soluble protein. We describe two procedures for detecting the stabilized protein in the soluble fraction of the samples. Whereas unbound proteins denature and precipitate at elevated temperatures, ligand-bound proteins remain in solution. The assay involves treatment of cells with a compound of interest, heating to denature and precipitate proteins, cell lysis, and the separation of cell debris and aggregates from the soluble protein fraction. The method allows studies of target engagement of drug candidates in a cellular context, herein exemplified with experimental data on the human kinases p38α and ERK1/2. Recently, we published a proof-of-principle study describing the implementation of thermal shift assays in a cellular format, which we call the cellular thermal shift assay (CETSA). Such assays have been used extensively on purified proteins in the drug discovery industry and in academia to detect interactions. Thermal shift assays are used to study thermal stabilization of proteins upon ligand binding. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
